Chemistry Chapter 7 Entry Test MCQs
51. The decomposition of ammonia to nitrogen and hydrogen on a tungsten filament at 800°C is independent of the concentration of ammonia at high pressures of ammonia. What is the order of the reaction with respect to ammonia?
a. Third
b. First
c. Zero
d. Second
52. Which of these graphs best describes the rate at which N2O4 decomposes to NO2 if the reaction is of first-order?
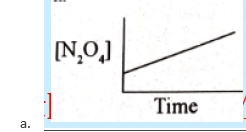
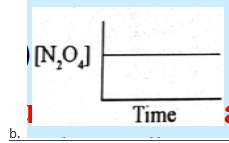

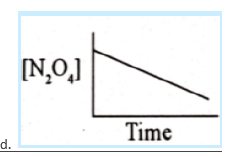
53. The rate law for the reaction A + 2B → products is Rate = K[A][B]2 when concentration of ‘B’ is increased from X to 3X by keeping the concentration of A constant. By what factor the rate of reaction will increase.
a. 6
b. 3
c. 27
d. 9
54. In a multistep reaction, the slowest step is:
a. always first step
b. Mechanism step
c. Enthalpy determining step
d. Rate determining step
55. In the reaction 2A + B → A2B, if the concentration of A is doubled and that of B is halved, then the rate of the reaction will
a. decrease 2 times
b. increase 2 times
c. remain the same
d. increase 4 times
Half life
56. For 3rd order reaction, half-life is inversely proportional to the initial concentration of reactants
a. Cube
b. Single
c. Raise to power four
d. Square
57. Which order of reaction obeys the relation t1/2 = 1/Ka
a. Third order
b. First order
c. Zero-order
d. Second order
58. For a first order reaction, the half life period is independent of:
a. Cube root of initial concentration
b. Initial concentration
c. Square root of final concentration
d. First power of final concentration
59. The half life of zero order reaction is:
a. Inversely proportional to initial concentration of reactants
b. Proportional to initial concentration of reactants
c. Graph with zero slope obtained
d. Independent of initial concentration of reactants
60. The % age of reactant left behind after four half time of chemical reactions one hour:
a. 6.25%
b. 3.125%
c. 25%
d. 12.5%
61. If the rate of decay of radio-active isotope decreases from 100gm to 25 gm after 24 hours. What is its half life:
a. 6 hours
b. 12 hours
c. 8 hours
d. 4 hours
62. The half life period for 3rd order reaction is:
a. [t1/2] α 1/a2
b. [t1/2] α 1/a0
c. [t1/2] α 1/a3
d. [t1/2] α 1/a1
63. Which of the following statements are correct about half life period?
(i) time required for 99.9% completion of a reaction is 100 times the half life period. (ii) time required for 75% completion of a 1st order reaction is double the half life of the reaction. (iii) it is proportional to initial concentration for zeroth order
a. ii and iii
b. i and iii
c. i only
d. i and ii
64. The half-life period for the reaction 2N2O5 ⇌2N2O4 + O2 is 24 minutes at 45°C. What will be the amount of N2O5 left behind after 96 minutes? When 10 g is starting amount.
a. 12.5g
b. 0.625g
c. 2g
d. 5g
65. If a first order reaction is 75% completed in 30 minutes, then how much time would have required for 50% completion of this reaction:
a. 20 min
b. 10 min
c. 7.5 min
d. 15 min
66. Incorrect statement among the following is
a. Rate of reaction is always positive quantity
b. Reactions have low activation energy values are fast
c. Half life of first order reaction is directly proportional to concentration
d. Increase in temperature increases rate of endothermic reactions
67. The half life of second order reaction is:
a. Inversely proportional to initial concentration of reactants
b. Directly proportional to initial concentration of reactants
c. Inversely proportional to square initial concentration of reactants
d. Directly proportional to square initial concentration of reactants
68. The half life period of a radioactive substance is 20 minutes. The percentage of the substance decayed in one hour.
a. 87.5%
b. 7.5%
c. 80%
d. 12.5%
Collision theory + Arrhenius
69. Graph between log k and (1/T) is linear of slope S. Hence Ea is
a. R/S
b. R & S
c. 2.303 RS
d. S/R
70. The Arrhenius equation accounts for the rate of a chemical reaction in terms of the
a. Physical states of the reactants
b. Concentration of reactants
c. Order of reaction
d. Activation energy
71. Collision theory explain:
a. Moleculerity of reaction
b. Rate of reaction
c. Order of reaction
d. All of these
72. Which of the equation is used to determine activation energy
a. Van der Waal equation
b. Henderson equation
c. general gas equation
d. Arrhenius equation
73. The value of activation energy is primarily determined by
a. Effective collision
b. Temperature
c. Chemical nature of reactants and products
d. Concentration of reactants
74. The unit of slope in the Arrhenius equation is _
a. Kelvin per mole
b. Kelvin
c. mole per Kelvin
d. Per Kelvin
75. When a graph is plotted between 1/T on X-axis and log k on y-axis a straight line is obtained with a negative slope which has two end in
a. III and IV quadrant
b. I and II quadrant
c. II and IV quadrant
d. II and III quadrant
